- Home
- Inclusions info
Inclusions types
Inclusions in Gemology
Inclusions studies
- Inclusions photo galleries
- Inclusions shop
- Shopping info
- Additional info
- Contact us
FTIR Spectroscopy
Introduction
FTIR spectroscopy is an improved form of infrared spectroscopy, which is a spectroscopic technique developed by mid XX century. As any other kind of spectroscopy, this procedure consists in the study of how the sample affects the light scattered or reflected by it (in this case infrared light). Infrared spectroscopy exploits the fact that molecules absorb specific frequencies that are characteristic of their structure, the so called resonant frequencies. These resonant frequencies depend on the vibrational modes of the molecules forming the material; each molecule absorbs several frequencies, the more complex the molecule is, the more different frequencies are absorbed. This is used in infrared spectroscopy to discover the composition of the sample.
The development of Fourier Transform infrared (FTIR) spectroscopy significantly increased the quickness, sensitivity and simplicity of this method, turning it into a widely used technique for identifying almost any material in a matter of seconds, thus allowing its use even in field analyses.
How it works
The traditional IR spectroscopy was dispersive type. In this technique, a laser beam was separated into different frequencies via dispersive prism or grafting. Each frequency light reached the sample (one at a time), and usually traversed it, or was reflected by it in some cases. In either case, a detector measured the amount of energy at each frequency coming from the sample, resulting into a spectrum. This procedure had important limitations. The first one was that it took some time to carry out the analysis, as illuminating the sample with many different frequencies was needed. Besides, most of the materials couldn’t reflect IR light well enough to perform the analysis, so the laser beam had to travel through them. For this to be possible, the sample had to be thinly laminated (not always possible) or pulverized, deteriorating the sample in both cases.
These problems were overcome with the invention of FTIR spectroscopy in the late 80s. This spectroscopy, rather than shining a monochromatic beam of light at the sample, shines a beam containing many frequencies of light at once, and measures how much of that beam is absorbed by the sample. Next, the beam is modified to contain a different combination of frequencies, giving a second data point. This process is repeated many times in a very short time.

Thermo Nicolet FTIR spectrometer
The reason why the beam does not have to be monochromatic is that this method utilizes a Michelson interferometer, which is an optical device that allows us to collect information about many different wavelengths simultaneously by splitting the beam by means of a certain configuration of mirrors, one of which is moved by a motor. As this mirror moves, each wavelength of light in the beam is periodically blocked, transmitted, blocked, transmitted, by the interferometer, due to wave interference. Different wavelengths are modulated at different rates, so that at each moment, the beam coming out of the interferometer has a different spectrum.
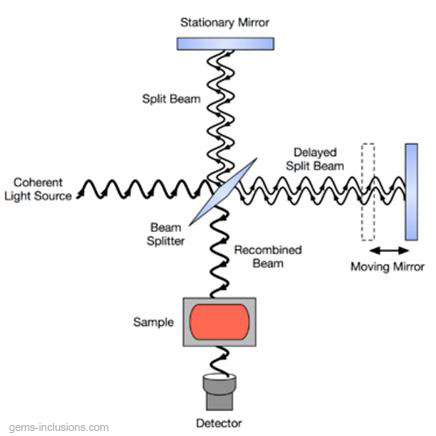
Schematic diagram of a Michelson interferometer, configured for FTIR
However, this information is unreadable, so a computer has to apply a Fourier Transform to it, which is a mathematical process that results into a plot representing the detected light intensity against the received light, usually in wavenumber. Wavenumber is a magnitude which represents the inverse of wavelength, commonly in cm⁻¹. This plot contains a set of absorption bands, corresponding to the frequencies absorbed by the sample, allowing us to find out its composition.
(spectrum picture)
On top of the highly increased speed of FTIR spectroscopy, the sensitivity is strongly improved and the signal-to-noise ratio is reduced in comparison to dispersive IR spectroscopy. Both these factors allow us to make analyses reflecting the beam into the sample in most of the materials. Furthermore, a very high spectral resolution can be achieved using FTIR. Although the advantages of the Michelson interferometer were known long ago, practical FTIR spectroscopes were not developed until 1987 because of the technical difficulty that supposed the rapidly and precisely moving mirror and the data processing capability needed for the Fourier Transform. As these difficulties were overcome, virtually all modern infrared spectrometers are FTIR instruments.
Pros and cons
FTIR spectroscopy has similar applications to Raman spectroscopy nowadays. Nevertheless, an important difference between them is that FTIR spectroscopy actually tells us the composition of the sample, while Raman only provides us a fingerprint that must be compared to a library to identify a material. This makes FTIR spectroscopy a very versatile technique, which can provide us accurate quantitative information about the substances present in a mixture or solution and the amount of each one, for example.
As it happens in Raman, an important advantage is the quickness of the analysis. The test itself takes only a couple of seconds. Depending on the spectrometer and the sample, this can be the total time of the analysis (in the case of handheld reflection spectrometers, f. e.), or it may need some sample preparation if the light has to go through the sample, and it is not transparent enough. The most common preparations for the analysis of solids are grinding the solid in a KBr matrix or dissolving the sample in a suitable solvent like CCl4 and CS2. Liquids and gases can be analyzed with this technology too, without much preparation.
Another favorable side of many FTIR spectrometers is the internal laser reference. The use of a helium neon laser as the internal reference in many FTIR systems provides an automatic calibration in an accuracy of better than 0.01 cm-1. This eliminates the need for external calibrations.
FTIR spectroscopy, both on transmission and reflection from the sample, is widely used in gemology, as it doesn’t damage the sample, it is fast, and the spectrometers are relatively cheap. It is suitable for inclusions study too because solids, liquids and gases can be analyzed, and the inclusions do not have to be on the surface of the stone, except if it is opaque to IR radiation.
Among its limitations, it has to be mentioned that this technique works almost exclusively on samples with covalent bonds and that it is hard to use in complex mixtures of organic substances.
References and links
- Griffiths, P.; de Hasseth, J.A. (2007). Fourier Transform Infrared Spectrometry.
- Nishikida, K.; Nishio, E.; Hannah, R.W. (1995). Selected applications of FT-IR techniques
- Banwell, C.N.; McCash, E.M. (1994). Fundamentals of Molecular Spectroscopy
- http://www.prenhall.com/settle/chapters/ch15.pdf
- http://www.newport.com/Introduction-to-FT-IR-Spectroscopy/405840/1033/content.aspx